GENDER
Male Brains Show A Chaotic Aging Process Compared To Female.
Published
5 years agoon
By
Adubianews
A study released Thursday in Stem Cell Reports focused on the brains of aging male and female mice. Blood vessels in male mouse brains, as opposed to female ones, showed more detrimental changes. The female brains seemed to have comparative protection.
These changes in brain blood vessels suggest potential differences in the way human male and female human brains age. Men, the study suggests, might have it worse.
“Everyone knows that neural stem cells decline with age,” says Sally Temple, the senior author of the study. Temple is the scientific director of the Neural Stem Cell Institute in Albany, New York.
“But now we have to say ‘in males’ because in females, we really didn’t see a significant loss. It was totally surprising,” Temple tells Inverse.
HERE’S THE BACKGROUND — Much of what we think we know about aging in the brain is based on studies of male mice.
“This is something we’re fighting all the time,” Temple says. “All the studies are done in males.”
With the help of a grant from the National Institute of Aging specifically for the study of sex differences, Temple teamed up with other researchers. This group includes Kristen Zuloaga, an associate professor of Neuroscience and Experimental Therapeutics at Albany Medical College.
Studies of mice have historically excluded females because of a misconception about their hormonal cycle, Zuloaga tells Inverse.
“They thought these changes in the hormone levels would lead to more variability within the female, so the females are messy,” Zuloaga says. “But it’s actually not the case at all.”
The researchers combined their interests in the study of sex differences in the brain with an inquiry into a specialized part of the brain that generates new neural cells called the “ventricular-sub ventricular zone.”
Ultimately, they wanted to observe how the brains of male and female mice differed when it came to generating new cells for the brain. The scientists didn’t expect to see those differences to be so vast.
DIGGING INTO THE SCIENCE — Here are the basics of how baby neurons, our most basic brain building blocks, are generally made: The brain’s stem cells, which can essentially generate any kind of specialized brain cell, are activated in a certain part of the brain called a “stem cell niche.” These cells create two kinds of intermediary cells, which give rise to young neurons. The neurons travel along pathways that follow blood vessels in the brain to their final destinations.
Blood vessels in the brain are of particular interest when understanding diseases of the brain because many of the problems observed in human brains, like a stroke or vascular dementia, come with changes in blood vessels.
WHAT THEY FOUND — Male and female mouse brains, as they aged, became marked by distinct differences in the blood vessel and neural stem cell systems. Female brain blood vessels became thicker but they weren’t crowded.
Male brain blood vessels, meanwhile, became denser, thinner, and more twisted. Females held on to their neural stem cells, while in males, they decreased and created fewer intermediary “progenitor” cells.
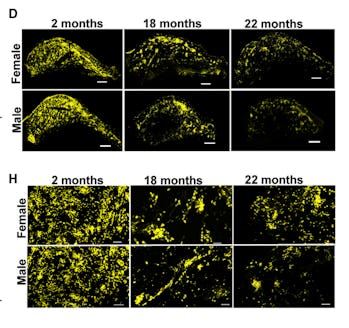
HOW THEY DID IT — The researchers dissected male and female mouse brains at three different ages and then stained them to differentiate between stem cells and blood vessels. They also analyzed the brain parts with a microscope in three dimensions, and created a three-dimensional computer reconstruction. This reconstruction let the team observe stem cells and blood vessels.
“Normally people will just take a thin slice, and they’ll be looking at just a tiny little piece of it,” Zuloaga says. “In this case, we’re able to look at the whole structure to see how the cells are kind of touching each other and where they’re located.”
The computer imaging allowed them to perform complex calculations to determine factors like density, diameter, and shape of the blood vessels over time.
WHY IT MATTERS — Because this study was done in mice, we can’t make conclusions about whether the same aging effects can be seen in human brains just yet. But the researchers say that there are a few signs that suggest at least some overlap in the patterns.
One encouraging sign is this particular area of the brain under the cortex plays a similar role in mice and humans. Its job is to harbor stem cells that then create new neural cells.
However, scientists are still debating whether or not this area is responsible for creating new brain cells in humans as the reach old age — which is what happens in mice.
Zuloaga also says that men are more likely than women to develop vascular dementia in old age, which is characterized by problems with blood flow to the brain. If what they saw in mice applied to humans, this discrepancy would make sense: the thinning and twisting of the blood vessels in the aging male mice would create problems for blood flow in the older brains of male mice but not females.
“WE DO HAVE TO PAY ATTENTION TO MALE-FEMALE DIFFERENCES THAT CAN AFFECT THINGS LIKE BLOOD VESSELS IN THE BRAIN…”
If the brains of human males and females undergo these same changes in aging as in the mice, it might change the way we understand and treat diseases of aging in males versus females.
“We do have to pay attention to male-female differences that can affect things like blood vessels in the brain, which are so critical for normal brain function,” Temple says. “But when they go wrong, they go wrong in stroke, they go wrong in vascular dementia, they go wrong in these diseases that affect so many people.”
WHAT’S NEXT — Animal models are critical for establishing potential sex differences in human brains, but researchers say the next step would be to study the brain differences in actual humans. Ideally, they would use some of the same 3-D imaging techniques that they did with the mice, Temple says.
She’s also conducting research with models called “organoids,” which are models of organs created with human stem cells used to study human disease.
The study team was also curious about further studies with mice that could examine the effects of other known conditions that affect brain health — like hypertension, diabetes, and menopause — on blood vessels and the generation of new brain cells.
But for now, there are many unknowns.
“If what we found in mice translates, then that I think it does really put a sort of question mark over this,” Temple says. “Males, in particular, need to be thinking about vascular health.”
But a number of lifestyle factors can encourage the continual creation of baby brain cells like exercising, sleeping enough, and keeping stress levels low.
“There are things you can do to boost neurogenesis and to boost your brain health,” Zuloaga says. “And they’re things that are pretty good for you anyway.”
You may like
-


Bombings in Iran: An African Policy Perspective on Global Risks and Economic Impact
-


Solomon Owusu Says Afenyo-Markin’s Apology Falls Short of Admitting False Recruitment Claims
-


Ghana Card Printing Resumes Nationwide After Technical Glitch — NIA Assures Public
-


Ablakwa Assures Protection for Ghanaians Amid Middle East Tensions
-


Kofi Adams Hints at Possible Andre Ayew Return for 2026 World Cup
-


Nana Agradaa Breaks Silence After Prison Release
-


Nana Agradaa Released After 9 Months in Prison
-


Aboagye: 24-Hour Economy Policy Still a Promise, Not Reality
-


Victoria Bright: Macro Gains Positive, But Structural Reforms Are Key

















